
Application Note
EarlyTox Cardiotoxicity Kit provides biologically relevant cardiotoxicity data earlier in the drug discovery process
- Study functional profiles in a biorelevant cardiotoxicity assay
- Minimize non-specific effect of dye on beat characteristics
- See temporal resolution with largest signal available
- Measure 384 samples in minutes rather than hours
Introduction
Assessing cardiotoxicity is important during early stages of drug discovery to eliminate potentially toxic compounds from further development. Highly predictive biologically relevant in vitro assays suitable for high throughput screening are critical to improve efficiencies and reduce high costs associated with compounds that fail during cardiac safety assessment. Stem cell-derived cardiomyocytes are especially attractive cell models because they express GPCRs and ion channels while demonstrating spontaneous mechanical and electrical activity similar to native cardiac cells.
The EarlyTox™ Cardiotoxicity Kit contains a novel calcium sensitive dye and a proprietary masking technology optimized for measuring changes in cytoplasmic calcium which are associated with cardiomyocyte beating. Characterization of beat patterns requires a short integration time. Because of the short camera exposure times, calcium binding to the dye within the cell produces a smaller signal than seen in standard calcium flux assays. This new formulation delivers a significantly brighter dye than other commercially available calcium indicators. As shown in Figure 1, the quench technology does not enter the cell, but reduces extracellular background fluorescence. Together these features help improve the signal window, providing more detail to the beat patterns.
The assay kit can be used for characterizing the impact of pharmacological compounds on peak frequency (BPM), peak amplitude and beat pattern and is designed to work with stem cell-derived cardiomyocytes or primary cardiomyocytes. Peak frequencies are determined from changes in intracellular Ca 2 concentration monitored by the EarlyTox Cardiotoxicity Dye. During a single beating cycle, Ca2 is released into the cytoplasm after the sarcoplasmic reticulum is stimulated. Calcium binds with troponin activating the sarcomere, and the cell contracts. Synchronously, there is an increase in fluorescent signal as dye binds to free calcium in the cytosol. Cellular relaxation occurs on removal of calcium from the cytosol by calcium uptake pumps of the sarcoplasmic reticulum and by calcium exchange with extracellular fluid. The fluorescent signal decreases as calcium concentration decreases. As the cycle repeats, additional fluorescent peaks synchronous with beating are observed.
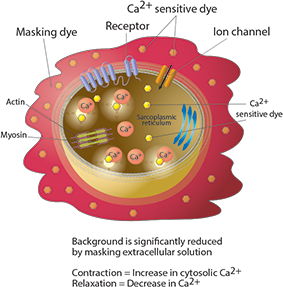
Figure 1. EarlyTox Cardiotoxicity Kit assay principle
Concentration-dependent modulation of calcium peak frequency & atypical patterns have been validated and characterized using the EarlyTox Cardiotoxicity Kit with cardiomyocytes on the FLIPR® Tetra System and analyzed with ScreenWorks® Peak Pro Software. Identified changes in parameters such as calcium peak frequencies, amplitude, peak width, rise and decay times enable the ability to redirect SAR or eliminate compounds from consideration that might otherwise be put forward to medicinal chemistry or to pre-clinical development. Additionally, this method may be used to enable the discovery of new compounds that affect the heart.
Materials and methods
In this assay, iCell Cardiomyocytes, human induced pluripotent stem cell-derived (iPSC) cardiomyocytes from Cellular Dynamics International were cultured according to their suggested protocol. Cells were plated in a 384-well black-walled clear plate 10–14 days prior to experiments. EarlyTox Cardiotoxicity Explorer Kit (R8210) dye loading buffer was warmed to 37°C to avoid slowing the beating of the cardiomyocytes upon addition. 25 μL dye was added to 25 μL cells in the plate. Each plate was incubated at 5% CO2, 37°C for two hours.
Three reference compounds that are included in the kit were studied to demonstrate a variety of effects upon the iPSC cardiomyocytes: propranolol, isoproterenol and sotalol. A compound plate containing concentration responses was made up at 5X final concentration in Component B buffer and warmed to 37°C prior to addition to minimize temperature effects upon the cells. After compound addition on the FLIPR Tetra System, cell beat rates and patterns can be monitored for up to 4–6 hours. Plates were read again 10 minutes post compound addition. Additional reads were taken at several time points up to four hours.
Results
Peak frequency and beat patterns are demonstrated for control and three reference compounds included in the EarlyTox Cardiotoxicity Kit as shown in Figures 2 through 5.
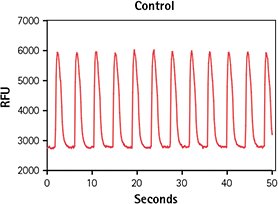
Figure 2. Untreated control iPSC cardiomyocytes. Calcium signal change in untreated iPSC cardiomyocytes incubated for 2 hours with EarlyTox Cardiotoxicity Dye indicated by calcium peaks.
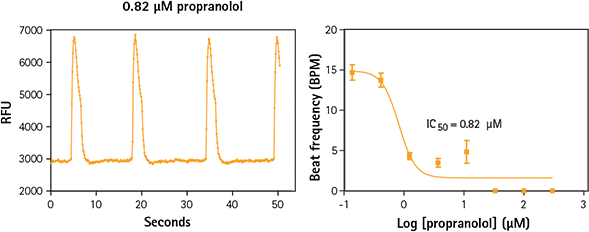
Figure 3. iPSC cardiomyocytes treated with propranolol exhibit a slower beat frequency. Propranolol is a non-selective blocker of ß1- and ß2-adrenergic receptors which inhibits the action of agonists of ß-adrenergic receptors. It slows peak frequency of calcium in iPSC cardiomyocytes.
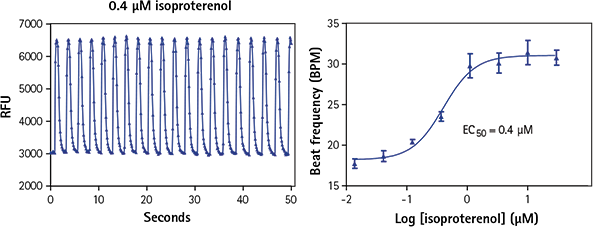
Figure 4. iPSC derived cardiomyocytes treated with isoproterenol show increased beat frequency. Isoproterenol is a non-selective ß-adrenergic agonist, which induces positive chronotropic and inotropic effects. It increases calcium peak frequency in iPSC cardiomyocytes.
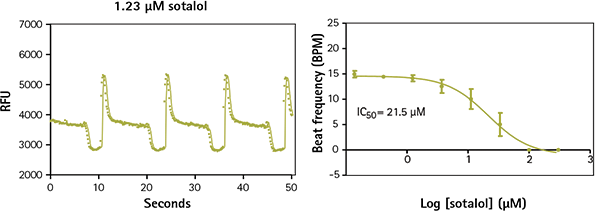
Figure 5. iPSC cardiomyocytes treated with sotalol show decreased beat frequency and altered beat pattern. Sotalol is a non-selective competitive ß-adrenergic receptor blocker that also inhibits hERG. In addition to slowing peak frequency of calcium in iPSC cardiomyocytes, it alters the normal pattern.
Calcium dye comparison
Multiple calcium indicators and their impact on the ability of the cells to beat, average peak amplitude, and peak frequency were compared over a four-hour period. Indicators included the EarlyTox Cardiotoxicity Kit, FLIPR ® Calcium 5 Assay Kit (Molecular Devices) and Fluo-4 Direct Kit (Invitrogen). Dyes were incubated for cell loading with iPSC cardiomyocytes according to vendor recommendations. A read was taken on the FLIPR Tetra System prior to- and at several time-points post-compound addition. Control wells were monitored in this experiment. As shown in Figures 6 and 7, overall peak frequency and signal amplitude were highest with the EarlyTox Cardiotoxicity Kit. As seen in Figure 8, there was no impact on the ability of the cells to beat (silenced) by the EarlyTox Cardiotoxicity Dye at four hours post compound addition. The FLIPR Calcium 5 Kit had silenced many wells, and all the wells were silenced at 90 minutes post-compound addition with the Fluo-4 Direct Kit.
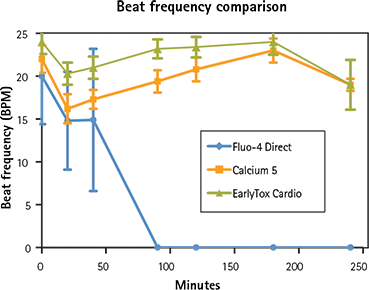
Figure 6. Beat frequency comparison Beat frequencies were recorded during a four hour period. EarlyTox Cardiotoxicity Dye had the highest beat frequency in all wells.
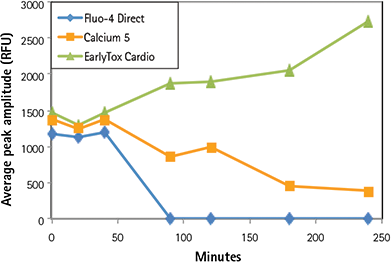
Figure 7. Impact of dye on average peak amplitude. Over time, EarlyTox Cardiotoxicity Dye had the highest average peak amplitude. Signal from the other dyes decreased over time.
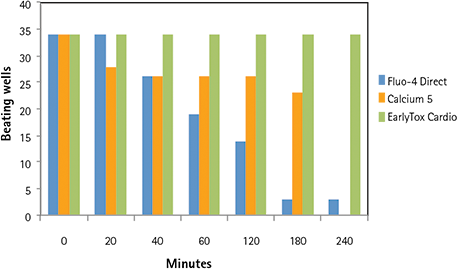
Figure 8. Impact of dye on cardiomyocyte beating over time. Thirty-four control wells were compared with each dye over a four-hour period. All of the EarlyTox Cardiotoxicity Dye wells remained beating up to four hours. Silenced wells occurred much earlier in the Fluo-4 Direct wells and the FLIPR Calcium 5 Kit wells and were nearly all gone by four hours.
Ordering information
(2) vials of Component A*
(1) bottle of dilution buffer (Component B)
(1) vial each of three reference compounds: isoproterenol, sotalol, propranolol
*
Each reagent vial (Component A) is sufficient for 1 plate (96-, 384-well). Each kit is sufficient for 2 plates.
(2) vials Component A
*
(1) bottle of dilution buffer (Component B)
(1) vial each of three reference compounds: isoproterenol, sotalol, propranolol
*
Each reagent vial (Component A) is sufficient for 5 plates (96-, 384-, 1536-well ). Each kit is sufficient for 10 plates.
Conclusions
The EarlyTox Cardiotoxicity Kit has been optimized to provide a robust, high throughput method for measuring the impact of pharmacological compounds on stem cell-derived cardiomyocytes. The optimized dye formulation provides the largest signal window and significantly reduced non-specific toxicity so there is minimal effect on beat rates, peak frequency or signal amplitude over time, unlike results from other commercially available dyes. When paired with the FLIPR Tetra System, the EarlyTox Cardiotoxicity Kit is a powerful tool for directing your drug design by allowing you to eliminate cardiotoxic compounds and identify potential cardiac drug candidates earlier in the drug discovery process.
Compatible with these Molecular Devices® systems

FLIPR® Tetra System.

SpectraMax® i3 Platform.

SpectraMax® Paradigm® Platform.

FlexStation® System.